1. Dau T, Wegner O, Mellert V, Kollmeier B. Auditory brainstem responses with optimized chirp signals compensating basilar-membrane dispersion. J Acoust Soc Am 2000;107:1530–40.


3. de Boer E. Auditory physics. Physical principles in hearing theory. 1. Phys Rep 1980;62:87–174.

4. Shore SE, Nuttall AL. High-synchrony cochlear compound action potentials evoked by rising frequency-swept tone bursts. J Acoust Soc Am 1985;78:1286–95.


5. Petoe MA, Bradley AP, Wilson WJ. On chirp stimuli and neural synchrony in the suprathreshold auditory brainstem response. J Acoust Soc Am 2010;128:235–46.


6. Cho SW, Han KH, Jang HK, Chang SO, Jung H, Lee JH. Auditory brainstem responses to CE-Chirp(R) stimuli for normal ears and those with sensorineural hearing loss. Int J Audiol 2015;54:700–4.


7. Cobb KM, Stuart A. Auditory brainstem response thresholds to airand bone-conducted CE-Chirps in neonates and adults. J Speech Lang Hear Res 2016;59:853–9.


8. Cebulla M, Stürzebecher E. Detectability of newborn chirp-evoked ABR in the frequency domain at different stimulus rates. Int J Audiol 2013;52:698–705.


9. Funasaka S, Ito S. Stimulus duration and waves of auditory brainstem response. Audiology 1986;25:176–83.


12. Dzulkarnain AAA, Noor Ibrahim SHM, Anuar NFA, Abdullah SA, Tengku Zam Zam TZH, Rahmat S, et al. Influence of two-electrode montages on the level-specific (LS) CE-Chirp auditory brainstem response (ABR) at multiple intensity levels. Int J Audiol 2017;56:723–32.


13. Kristensen SG, Elberling C. Auditory brainstem responses to levelspecific chirps in normal-hearing adults. J Am Acad Audiol 2012;23:712–21.


14. Cargnelutti M, Cóser PL, Biaggio EPV. LS CE-Chirp(R) vs. Click in the neuroaudiological diagnosis by ABR. Braz J Otorhinolaryngol 2017;83:313–17.


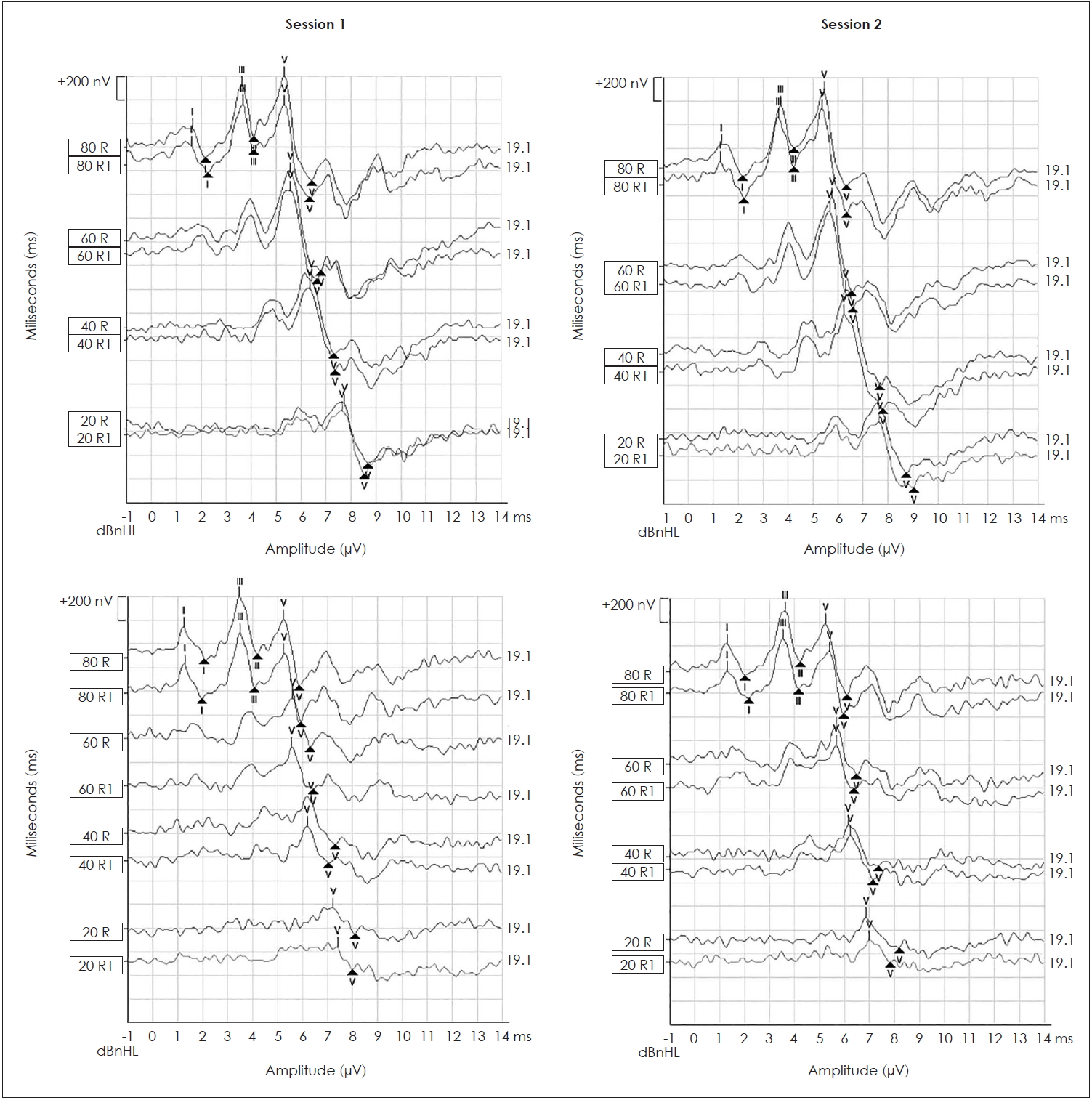
15. Weir JP. Quantifying test-retest reliability using the intraclass correlation coefficient and the SEM. J Strength Cond Res 2005;19:231–40.


16. Beattie RC, Zipp JA, Schaffer CA, Silzel KL. Effects of sample size on the latency and amplitude of the auditory evoked response. Am J Otol 1992;13:55–67.


17. Dzulkarnain AAA, Buyong AS, Sulaiman NH. Intra-subject variability in the auditory brainstem response using a vertical montage recording. Speech Lang Hear 2014;17:160–7.

18. Oyler RF, Lauter JL, Matkin ND. Intrasubject variability in the absolute latency of the auditory brainstem response. J Am Acad Audiol 1991;2:206–13.

19. vd Drift JF, Brocaar MP, von Zanten GA, Lamoré PJ. Inaccuracies in the measurement of auditory brainstem response data in normal hearing and cochlear hearing loss. Audiology 1988;27:109–18.


20. Cobb KM, Stuart A. Test-retest reliability of auditory brainstem responses to chirp stimuli in newborns. Int J Audiol 2014;53:829–35.


21. Edwards RM, Buchwald JS, Tanguay PE, Schwafel JA. Sources of variability in auditory brain stem evoked potential measures over time. Electroencephalogr Clin Neurophysiol 1982;53:125–32.


22. Tusa RJ, Stewart WF, Shechter AL, Simon D, Liberman JN. Longitudinal study of brainstem auditory evoked responses in 87 normal human subjects. Neurology 1994;44(3 Pt 1):528–32.


23. Lauter JL, Loomis RL. Individual differences in auditory electric responses: comparisons of between-subject and within-subject variability. I. Absolute latencies of brainstem vertex-positive peaks. Scand Audiol 1986;15:167–72.


24. Elberling C, Wahlgreen O. Estimation of auditory brainstem response, ABR, by means of Bayesian inference. Scand Audiol 1985;14:89–96.


25. Rowe MJ 3rd. Normal variability of the brain-stem auditory evoked response in young and old adult subjects. Electroencephalogr Clin Neurophysiol 1978;44:459–70.


27. Lauter JL, Loomis RL. Individual differences in auditory electric responses: comparisons of between-subject and within-subject variability. II. Amplitude of brainstem Vertex-positive peaks. Scand Audiol 1988;17:87–92.


28. Lauter JL, Karzon RG. Individual differences in auditory electric responses: comparisons of between-subject and within-subject variability. V. Amplitude-variability comparisons in early, middle, and late responses. Scand Audiol 1990;19:201–6.


30. Elberling C, Don M. Detecting and assessing synchronous neural activity in the temporal domain (SNR and response detection). In: Burkard RF, Eggermont JJ, Don M. editors. Auditory evoked potentials: basic principles and clinical application. 1st ed. Baltimore: Lippincott, Willimas & Wilkins;2007. p.102–23.