 |
 |
- Search
| J Audiol Otol > Volume 27(2); 2023 > Article |
|
Abstract
Background and Objectives
Subjects and Methods
Results
Acknowledgments
Notes
Author Contributions
Conceptualization: Kapil Sikka, Rabia Monga. Data curation: Anupam Kanodia, Yash Verma. Formal analysis: Anupam Kanodia, Aanchal Kakkar. Funding acquisition: Kapil Sikka. Investigation: Hitesh Verma, Aanchal Kakkar. Methodology: Aanchal Kakkar, Diya Roy. Project administration: Alok Thakar, Kapil Sikka. Resources: Kapil Sikka, Aanchal Kakkar. Software: Anupam Kanodia. Supervision: Alok Thakar. Validation: Chirom Amit Singh, Deepali Jain. Visualization: Aanchal Kakkar, Diya Roy, Deepali Jain. Writingã original draft: Anupam Kanodia, Aanchal Kakkar. Writingãreview & editing: Aanchal Kakkar, Kapil Sikka. Approval of final manuscript: all authors.
Fig.ô 1
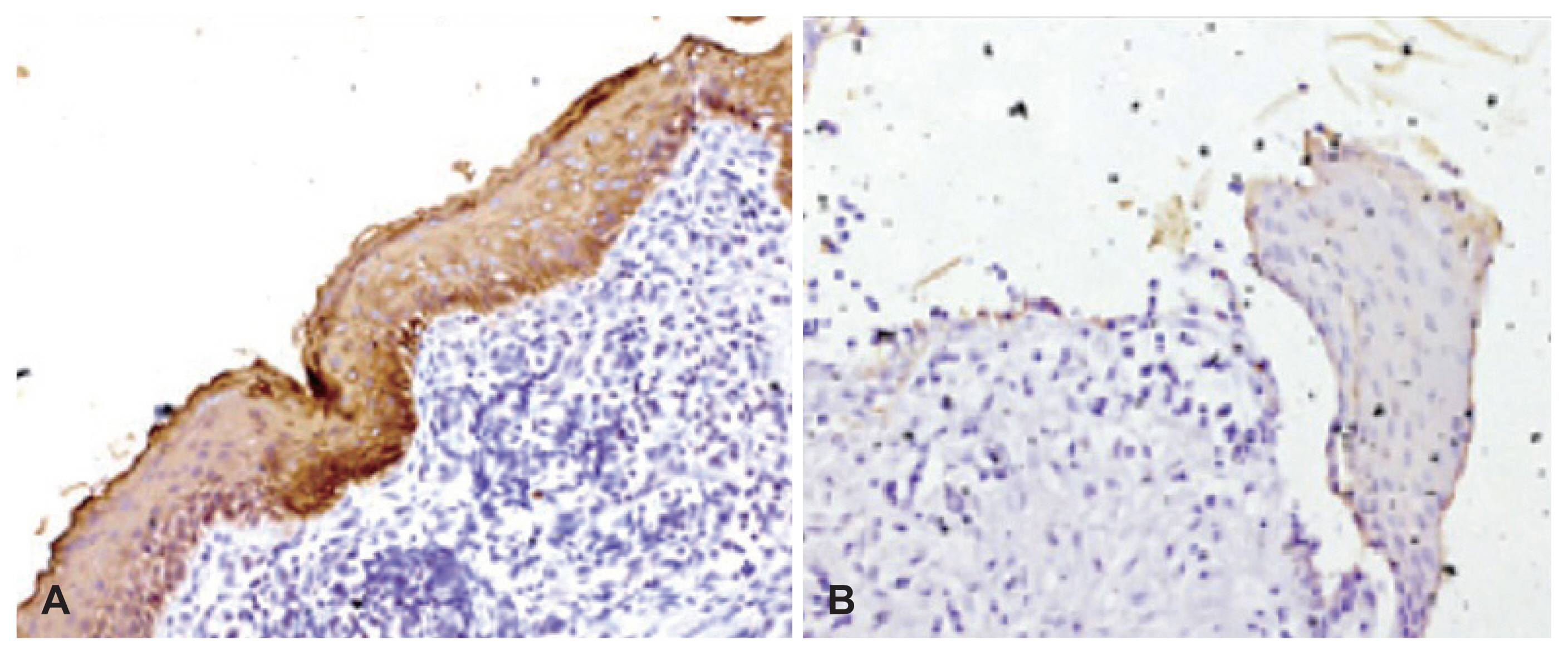
Fig.ô 2
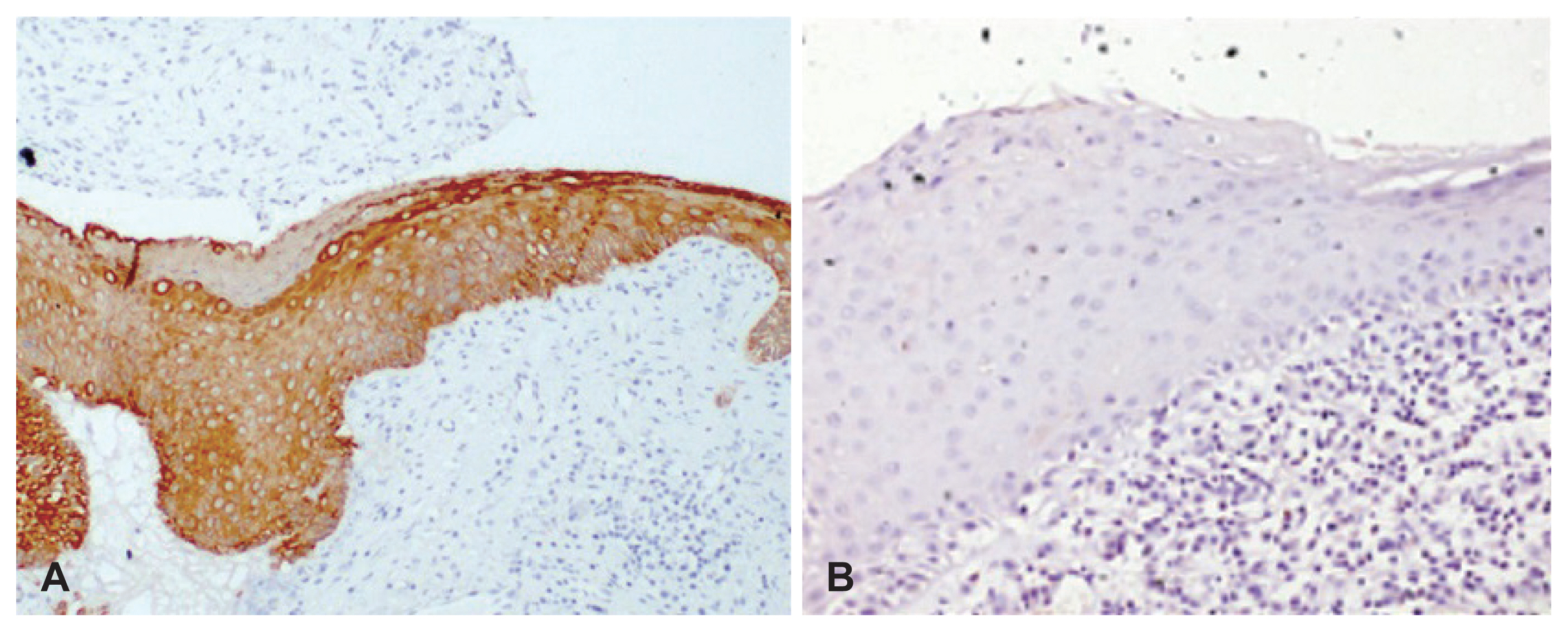
Fig.ô 3

Fig.ô 4
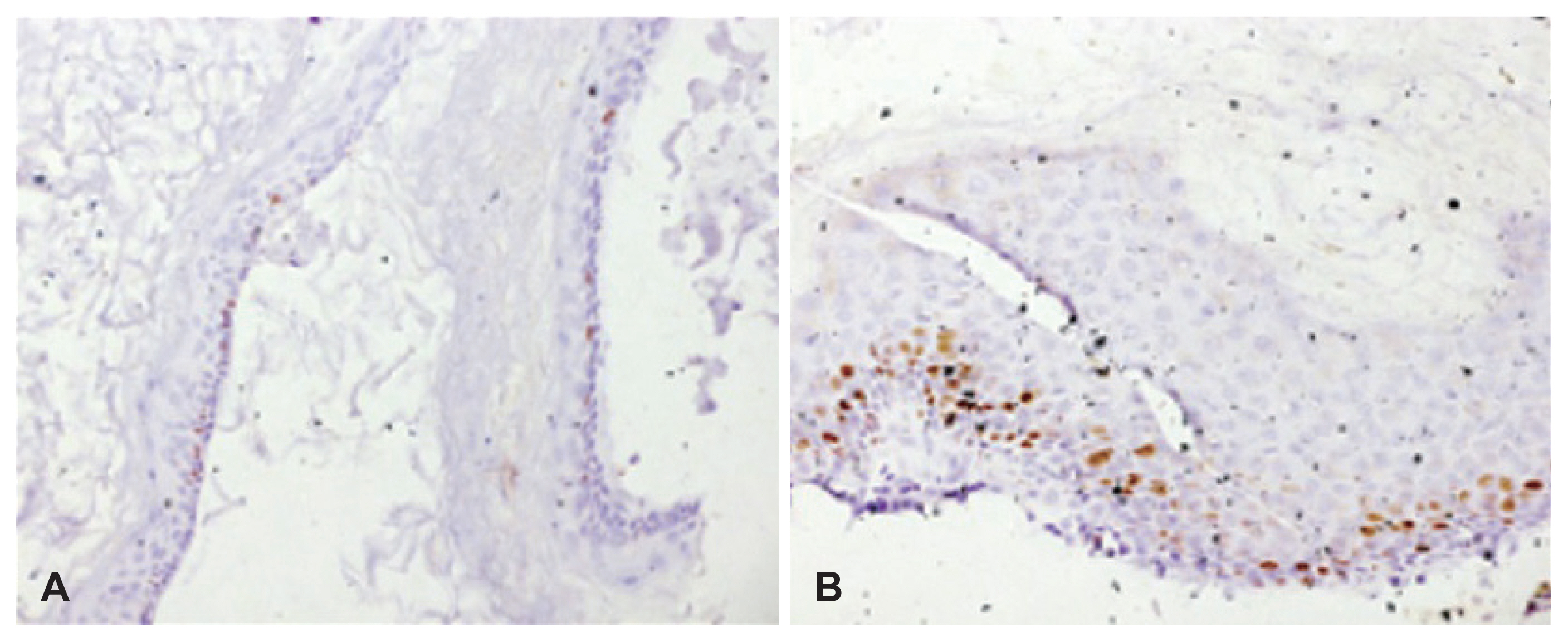
Tableô 1
Tableô 2
| Stage | 34ûe12 immunopositivity | CK13 immunopositivity | CK17 immunopositivity |
|---|---|---|---|
| Stage I (10 specimens) | 10 | 6 | 10 |
| Stage II (27 specimens) | 23 | 23 | 23 |
| Stage III (4 specimens) | 4 | 3 | 3 |
| Stage IV (0 specimens) | - | - | - |
Tableô 3
For 34ûe12, two subgroups were full-thickness expression and negative staining group (negative staining+basal pattern). For CK17, specimens were divided into suprabasal group (which included suprabasal pattern, full-thickness pattern and suprabasal+superficial pattern), superficial only group, and negative group. For CK13, specimens were divided as full-thickness expression group, patchy expression group (suprabasal, basal or both), and negative staining group. For Ki67 stain, the specimens were divided as mild expression group (1ã3 layers), high expression group (4ã5 layers), and negative staining group. IHC, immunohistochemistry







